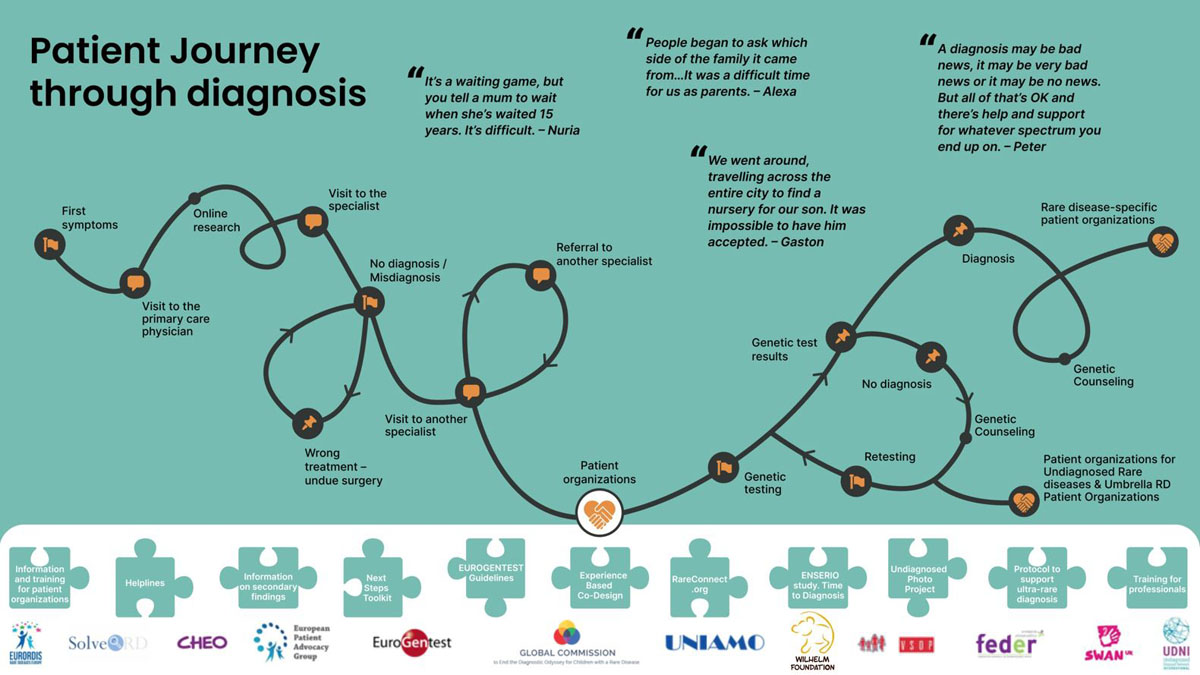
Journey infographic
Click on the logos and the puzzle pieces to learn more!
EURORDIS-Rare Diseases Europe

EURORDIS-Rare Diseases Europe is a unique, non-profit alliance of almost 1000 rare disease patient organisations from morre than 70 countries that work together to improve the lives of the 30 million people living with a rare disease in Europe. By connecting patients, families and patient groups, as well as by bringing together all stakeholders and mobilising the rare disease community, EURORDIS strengthens the patient voice and shapes research, policies and patient services.
Solve RD
Solve-RD - solving the unsolved rare diseases is a research project funded by the European Commission for five years (2018-2022). It echoes the ambitious goals set out by the International Rare Diseases Research Consortium (IRDiRC) to deliver diagnostic tests for most rare diseases by 2020. The current diagnostic and subsequent therapeutic management of rare diseases is still highly unsatisfactory for a large proportion of rare disease patients – the unsolved RD cases. For these unsolved rare diseases, we are unable to explain the etiology responsible for the disease phenotype, predict the individual disease risk and/or rate of disease progression, and/or quantitate the risk of relatives to develop the same disorder.
ePAG
EURORDIS wants patient organisations to participate in European Reference Network (ERN) decision-making processes and is supporting its membership to ensure a democratic process of patient representation.
For this reason, EURORDIS has developed a European Patient Advocacy Group (ePAG) for each ERN disease grouping. ePAGs will bring together elected patient advocates and affiliated organisations who will ensure that the patient voice is heard throughout the ERN development process.
EuroGentest
EuroGentest is a project funded by the European Commission to harmonize the process of genetic testing, from sampling to counseling, across Europe. The ultimate goal is to ensure that all aspects of genetic testing are of high quality thereby providing accurate and reliable results for the benefit of the patients.
Global Commission
The Global Commission to End the Diagnostic Odyssey for Children with a Rare Disease is a multi-disciplinary group of experts committed to accelerating the time to diagnosis. The Global Commission has developed an actionable roadmap to guide the rare disease field in shortening the multi-year journey that patients endure to receive an accurate diagnosis, which can be the key to a longer, healthier life.
UNIAMO
UNIAMO Italian Federation of Rare Diseases is the representative body of the community of people with rare diseases. It has been working for over 20 years for the protection and defense of the rights of people with rare diseases and their families, it has over 150 affiliated associations in continuous growth.
Wilhelm Foundation
Wilhelm Foundation is a non-profit organization that work internationally to improve the diagnostics of undiagnosed patients around the world. The mission is that all undiagnosed patients get a diagnosis by working with specialists and researchers in diagnostics to solve the undiagnosed diseases which are not discovered yet in the medical literature. Co-founder of UDNI and the International Conferences on Rare and Undiagnosed Diseases. Located in Sweden, with a parent support group, family camps, parent’s weekends and grandparent’s support group.
VSOP
VSOP is the Dutch national patient umbrella organization for rare and genetic disorders with a membership of approximately 87 disease-specific patient and parent organizations. Its mission is to improve the quality of life of patients with (ultra-)rare diseases and their families.
FEDER
FEDER is an umbrella alliance that brings together more than 370 patient organizations.
Its mission is to represent and defend the rights of people with RD or with suspected diagnosis, to improve their quality of life.
SWAN
SWAN UK (syndromes without a name) is a support network for familes of children and young adults with undiagnosed genetic conditions. We are run by the charity Genetic Alliance UK. We offer suport, information and run events to bring families together accross the UK. We want it recognised that being undiagnosed is not always a temporary stage; the genetic cause of some conditions may never be known.
UDNI
The UDNI involves centers with internationally recognized expertise, and its scientific resources and know-how aim to fill the knowledge gaps that impede diagnosis. Consequently, the UDNI fosters the translation of research into medical practice. Active patient involvement is critical; the Patient Advisory Group is expected to play an increasing role in UDNI activities.
Rareconnect.org
Rareconnect.org is a safe, easy to use platform where rare disease patients, families and patient organizations can develop online communities and conversations across continents and languages. RareConnect partners with the world's leading rare disease patient groups to offer global online communities allowing people to connect around issues which affect them while living with a rare disease.The community for undiagnosed diseases in Rareconnect.og currently has 561 members and 20 patient organizations.
Undiagnosed Photo Project
The goal with Wilhelm Foundation's photo project the UNDIAGNOSED is to give the ones who suffer from an undiagnosed disease another chance to a diagnosis by presenting their photograph at a photo exhibition at the International Conference on Rare and Undiagnosed Diseases. The Foundation also distributes a booklet of the photographs and related medical information to all specialists at world congresses to increase the chance of diagnosis for the Undiagnosed.The participants in the project are those who have an undiagnosed disease and undergone an investigation on an Undiagnosed Diseases Program (UDP) and are nevertheless undiagnosed.
Next Steps Toolkit
The goal of the Next Steps toolkit is to help patients that are exiting UDP programs navigate the next step in the journey with or without a diagnosis. The toolkit aims to achieve this by gathering resources and providing this information to UDP healthcare providers so that they can give patients access to these resources.
Helplines
People living with a rare disease are frequently faced with a critical lack of information and support. Upon hearing their diagnosis, their first point of contact is often the local patient organisation for their disease.
If a patient organisation does not exist in their area or cannot supply the correct, validated information, the patient is left feeling isolated.
Rare disease help lines offer social, psychological and information solutions to all of these needs. Available help lines are listed below.
EURORDIS promotes, networks, trains and advocates on behalf of rare disease help lines. In order to create a service that provides quality information and support, patient organisations need guidance and assistance. The European Network of Rare Diseases Help Lines provides this guidance.
EUROGENTEST Guidelines
Best practice guidelines for genetic testing are recommendations describing techniques or methods to perform a specific test in the best way possible. It provides professionals with a standard to perform the tests. This promotes uniformity in testing and results in a higher quality in terms of accuracy of results and interpretation.
These guidelines are usually drafted after best practice meetings with experts from the field. EuroGentest has actively participated in and organized many of these meetings in molecular genetics, biochemical genetics and (molecular) cytogenetics.
ENSERIO
The ENSERio Study investigated patients’ experiences and expectations regarding access to diagnosis, treatment healthcare and social services. It is based on interviews, discussion groups and a survey conducted amongst 715 People Living With Rare Diseases (PLWRD) across Spain.
This update was powered by FEDER and CREER dependent on the IMSERSO during the years 2016-2017. This time the sample of the first study was duplicated obtaining a total amount of 1576 questionnaires, 14 interviews and 8 focus groups. This sample came from FEDER, CREER and SpainRDR (Spanish Rare Diseases Registries Research Network). The methodology used was the same as in the first ENSERio study in order to make it possible to compare the results and analyze the possible trends and developments with the first reference work.
Information on secondary findings
AnDDI-Rares organization prepared a video giving information on secondary findings from genome sequencing.
https://www.youtube.com/watch?v=Z1k3xN5rKvU
